An unusual cobalt(ii)-based single-walled metal–organic nanotube†
Abstract
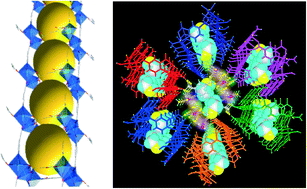
Three rare Co(II)–organic frameworks were synthesized by tuning the relative reactant ratios of CoII : 2,4-H2pydc (2,4-H2pydc = 2,4-pyridinedicarboxylic acid) and introducing different structure-directing amines under hydrothermal conditions. Compound {[Co3(2,4-pydc)3(EtOH)(H2O)3]·EtOH}n (1), which was prepared using a 1 : 1 ratio of CoII : 2,4-H2pydc, consisted of a tricobalt(II) cluster that was employed to form a two-side-open box and these boxes were further self-assembled into a rare single-walled metal–organic nanotube (MONT). When the ratio was adjusted to 2 : 1 and accompanied with 4,4′-dipyridyl-piperazine (dpyp), the formation of zigzag chains of {[Co4(2,4-pydc)4(H2O)10]}n (2) with a tetracobalt(II) cluster unit occurred, finally these chains were linked into a 2D sheet with a 44-sql topology. However, compound {[Co(2,4-pydc)2(H2O)2]·[Co(dpyp)(H2O)4]}n (3) was synthesized under similar reaction conditions as for 2 except that triethylamine was added. 3 contained the polymeric chains of {[Co(dpyp)·(H2O)4]2+}n and the monocobalt complex [Co(2,4-pydc)2(H2O)2]2−, which were cooperatively connected via hydrogen bonding to form a 2D layer with a 44-sql topology. A pseudo-merohedral twinning law was applied to the twin case during the X-ray structural analysis of 2 that facilitated the R1 value to drop drastically.

 Please wait while we load your content...
Please wait while we load your content...