A 3D MOF showing unprecedented solvent-induced single-crystal-to-single-crystal transformation and excellent CO2 adsorption selectivity at room temperature†
Abstract
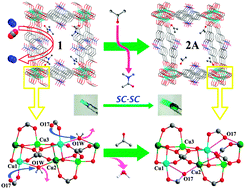
A water stable porous 3D metal–organic framework, [Cu3L2(μ3-OH)2(μ2-H2O)]·2DMA (1, mother crystal, H2L = 2,2′-dinitrobiphenyl-4,4′-dicarboxylic acid, DMA = N,N-dimethylacetamide), shows unprecedented irreversible solvent-induced substitutions of bridging aqua ligands and guest-exchanges in single-crystal-to-single-crystal (SCSC) transformations at room temperature (RT), producing quantitatively three daughter crystals, [Cu3L2(μ3-OH)2]·2S (2: 2A, S = acetone; 2B, S = 2-propanol; 2C, S = 2-butanol), which exhibit reversible interconversion by guest-exchanges at RT in SCSC transformations. MOF 1 shows excellent separation selectivity (128) of CO2/N2 at RT and is a better sorbent of micro-solid-phase extraction (μ-SPE) than currently known benchmark ZIF-8.

 Please wait while we load your content...
Please wait while we load your content...