LaF3 nanoparticle-assisted sensitive detection of protein kinase activity†
Abstract
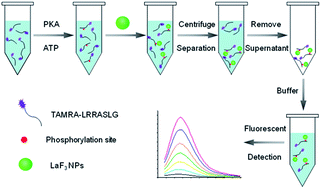
A fluorescent protein kinase assay is developed based on the selective recognition ability of LaF3 nanoparticles (LaF3 NPs) towards a phosphorylated peptide substrate. In this study, protein kinase can catalyze the phosphorylation of the fluorescently labeled substrate peptide, which may then bind specifically on the surface of LaF3 NPs through the strong interaction between La3+ and the phosphate group while the unphosphorylated peptide will not. Therefore, fluorescent phosphopeptides are enriched on the LaF3 NP surfaces in correlation with the kinase activity. After isolation of the unbound peptides via centrifugation, the fluorophore-anchored LaF3 NPs can be easily re-dispersed in aqueous buffer to form a stable colloid solution. By monitoring the fluorescence signal of such colloid solution, the activity of protein kinase A (PKA), a proof-of-concept protein kinase, can be easily determined from 0.0006 U μL−1 to 0.2 U μL−1 with a detection limit of 0.0002 U μL−1. By combining with the synchronous fluorescence technique, the proposed assay is feasible for simultaneous detection of multiple protein kinases. Furthermore, this simple method is also successfully applied to the PKA inhibition assay, showing great potential in the study of cellular signal transduction and drug discovery.

 Please wait while we load your content...
Please wait while we load your content...