Quantification and pharmacokinetics of astragaloside II in rats by rapid liquid chromatography-tandem mass spectrometry
Abstract
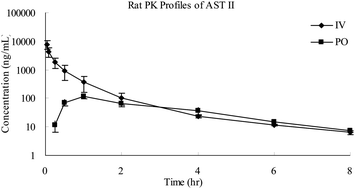
This study first describes the development of a rapid and accurate high-performance liquid chromatography-tandem mass spectrometry (LC-MS/MS) assay for the quantification of astragaloside II (AST II) in rat plasma. The assay involved a simple protein precipitation (PPT) step with methanol–acetonitrile (50 : 50, v/v) and a gradient elution using a mobile phase consisting of water containing 0.1% formic acid and acetonitrile containing 0.1% formic acid. Chromatographic separation was successfully achieved on an Agilent Zorbax XDB C18 column (2.1 mm × 50 mm, 3.5 μm) with a flow rate of 0.50 mL min−1. Multiple reaction monitoring (MRM) was based on the transitions of m/z = 827.3 → 143.2 for AST II and 386.3 → 122.3 for buspirone (IS). The assay was validated to demonstrate the specificity, linearity, recovery, accuracy, precision and stability. The lower limit of quantification (LLOQ) was 5.0 ng mL−1 in 50 μL of rat plasma. The developed and validated method has been successfully applied to the quantification and pharmacokinetic study of AST II in rats after intravenous and oral administration of AST II. The oral absolute bioavailability (F) of AST II was calculated to be 0.79 ± 0.16% with an elimination half-life (t1/2) value of 1.92 ± 0.30 h, suggesting its poor absorption and/or strong metabolism in vivo.

 Please wait while we load your content...
Please wait while we load your content...