A nitric oxide quantitative assay by a glyceraldehyde 3-phosphate dehydrogenase/phosphoglycerate kinase/firefly luciferase optimized coupled bioluminescent assay†
Abstract
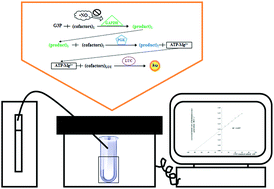
A novel optimized coupled bioluminescent assay for nitrogen monoxide free radical (nitric oxide, ˙NO), an important environmental and physiological molecule, is presented. The method is based on the reaction catalyzed by glyceraldehyde 3-phosphate dehydrogenase (GAPDH), whose product is used as a substrate for phosphoglycerate kinase (PGK), generating adenosine 5′-triphosphate (ATP), which is an essential cofactor for the firefly luciferase bioluminescent reaction. Inhibition of GAPDH by ˙NO hampers the coupled reactions, leading to a depletion of ATP and hence a decrease in the bioluminescent signal. Using diethylamine NONOate (DEA-NONOate) as the ˙NO donor, the assay was optimized through statistical experimental design methodology, namely Plackett–Burman (screening) and Box–Behnken (optimization) designs. The optimized method requires 5 μL of sample per tube in a final reaction volume of 100 μL. It is linear in the range from 10 to 100 nM of ˙NO, with limits of detection and quantitation of 4 and 15 nM, respectively. Limitations in its application to biological samples, together with approaches to solve them, are discussed using human whole saliva and microalgae culture medium as examples.

 Please wait while we load your content...
Please wait while we load your content...