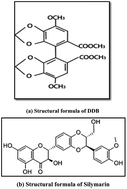
Derivative spectrophotometric and micellar high performance liquid chromatographic methods for simultaneous determination of silymarin and dimethyl-4,4′-dimethoxy-5,6,5′,6′-dimethylene dioxybiphenyl-2,2′-dicarboxylate (DDB) in raw materials and capsules
Abstract
First derivative ultraviolet spectrophotometric and micellar high performance liquid chromatographic assay methods were developed and validated for the simultaneous determination of silymarin and dimethyl-4,4′-dimethoxy-5,6,5′,6′-dimethylene dioxybiphenyl-2,2′-dicarboxylate (DDB) in the pure form and in combined capsules. The first derivative (D1) was performed by measuring the amplitudes at 279 and 255 nm for silymarin and DDB, respectively. The calibration graphs were linear in the range of 3–40 μg mL−1 for silymarin and 3–30 μg mL−1 for DDB. An isocratic reversed phase chromatographic assay method was also developed for the determination of both drugs, a C18 column (250 mm × 4.6 mm i.d., 5 μm particle size) was used with a mobile phase composed of 0.17 M sodium dodecyl sulphate, 12% butanol and 0.3% triethylamine, all prepared in 0.02 M phosphoric acid and adjusted to pH 3.5. The flow rate was 1 mL min−1. The effluents were monitored at 288 nm and floctafenine was used as the internal standard. Plots of the peak area ratios of each drug to floctafenine were found to be linear in the range of 1–15 μg mL−1 for silymarin and 1–12 μg mL−1 for DDB. The proposed methods were validated according to ICH guidelines. The methods were successfully applied in the estimation of silymarin and DDB in their combined capsules dosage form.

 Please wait while we load your content...
Please wait while we load your content...