Development and validation of an HPLC-UV method for quantifying nevirapine and its main phase I metabolites in human blood
Abstract
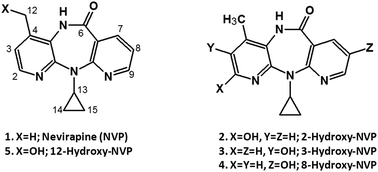
Nevirapine (NVP) is a widely used drug for the treatment of HIV-infection, which is associated with severe toxic events dependent on its biotransformation. Thus, the availability of analytical methods for the biomonitoring of NVP and its main metabolites is of utmost importance. In this paper we report the development and validation of a reversed phase HPLC-UV method for the quantification of NVP and its main phase I metabolites in human plasma. The method was validated over a range of 10–2500 ng mL−1 for the phase I metabolites of NVP and 10–10 000 ng mL−1 for NVP. The coefficients of variation (CV) of the average back-calculated concentrations were lower than 9% and the LLOQ was 10 ng mL−1 for each analyte. The accuracy and precision of the method were acceptable. There was no significant interference from plasma components or from other typically co-administered antiretroviral drugs and the mean recovery was 94%. This method represents an inexpensive, sensitive, accurate and precise alternative to mass spectrometry-based biomonitoring of NVP.

 Please wait while we load your content...
Please wait while we load your content...