Adsorption of folic acid, riboflavin, and ascorbic acid from aqueous samples by Fe3O4 magnetic nanoparticles using ionic liquid as modifier
Abstract
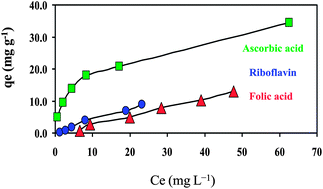
In this paper, nanoparticles of Fe3O4 as well as its binary mixtures with ionic liquid 1-hexyl-3-methylimidazolium bromide (IL–Fe3O4) were prepared and used for adsorption of folic acid, riboflavin and ascorbic acid. The mean size and the surface morphology of both nanoparticles were characterized by transmission electron microscopy, X-ray diffraction, thermogravimetric analysis and Fourier transform infrared spectroscopy techniques. The pH of the point of zero charge (pHpzc) of both Fe3O4 and IL–Fe3O4 were obtained based on the experimental curves corresponding to the immersion technique. Experimental results were obtained under the optimum operational conditions of: 40 mg of nanoparticle and contact time of 10 minutes with an initial concentration of the vitamins of 20 mg L−1. The isotherm evaluations revealed that the Freundlich model attained better fits to the equilibrium data than the Dubinin–Radushkevich model. The maximum obtained adsorption capacities of folic acid, riboflavin and ascorbic acid were 22.5, 4.8 and 6.9 mg per gram of adsorbent, respectively. The adsorption capacities of the vitamins were found to be dependent on their chemical structures, pH of the solution and temperature. The applicability of pseudo-first order and pseudo-second order kinetic models was estimated on the basis of comparative analysis of the corresponding rate parameters, equilibrium adsorption capacity and correlation coefficients. Furthermore, both adsorption processes of folic acid and ascorbic acid were endothermic and vitamins could be desorbed from IL–Fe3O4 nanoparticles by using NaCl solution at pH 3.0 for recycling the nanoparticles.

 Please wait while we load your content...
Please wait while we load your content...