Stability improvement of Prussian blue in nonacidic solutions via an electrochemical post-treatment method and the shape evolution of Prussian blue from nanospheres to nanocubes
Abstract
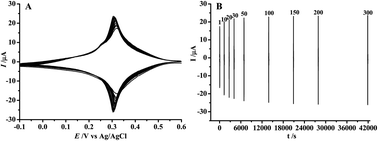
In this study we report the improved stability of Prussian blue (PB) films in neutral and alkaline solutions via an electrochemical post-treatment procedure. In this procedure, PB films synthesized via the electroless deposition method at glassy carbon electrodes were continuously cycled for 300 cycles (∼12 h) between the potential limits of +0.6 and −0.1 V (vs. Ag/AgCl) at a scan rate of 10 mV s−1 in 2.0 M KCl solution of pH 3. It was found that after such electrochemical post-treatment the PB films exhibit dramatically improved stability in neutral and alkaline solutions. Control experiments confirmed that the longer potential cycling time and the higher KCl concentration are indispensable. In addition, interestingly, it was found that after the successive potential cycling treatments the shape of PB nanoparticles evolves from a sphere into a cube as well as a small amount of a rectangle, indicating the electrochemically induced shape evolution of nanocrystals. The electrochemical post-treatment procedure proposed here could be useful for the development of PB-based devices with improved stability, for instance, biosensors, biofuel cells, etc.

 Please wait while we load your content...
Please wait while we load your content...