Multi-addressable molecular switches based on a new diarylethene salicylal Schiff base derivative†
Abstract
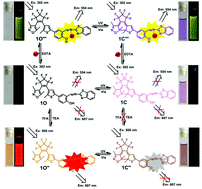
A novel photochromic diarylethene with a salicylidene Schiff base substituent has been synthesized and characterized. The diarylethene could be used as a multi-addressable fluorescence switch when triggered by light,

 Please wait while we load your content...
Please wait while we load your content...