Effect of anions in Au complexes on doping and degradation of graphene†
Abstract
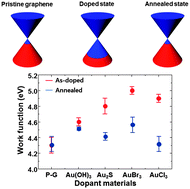
In this study, the role of anions in Au complexes was investigated as a dopant for graphene. Au(OH)3, Au2S, AuBr3, and AuCl3 were used as dopants. The average sheet resistance of the graphene decreased from 950 Ω sq−1 to 820, 600, 530, and 300 Ω sq−1 and the work function increased from 4.3 eV to 4.6, 4.8, 5.0 and 4.9 eV with Au(OH)3, Au2S, AuBr3, and AuCl3 dopants, respectively. However, thermal annealing of graphene increased the sheet resistance and decreased the work function. Furthermore, the G and 2D bands in the Raman spectra were recovered and the Au3+ peak in the Au 4f spectra decreased after thermal annealing, indicating the aggregation of metal particles. These results suggested that the degree of doping was related to the electronegativity of the anion in the Au complex and degradation of graphene was related to the bond strength between the Au cation and the counter anion. Therefore, anions with a high electronegativity and high bond strength are adequate for use as a p-type dopant in graphene.

 Please wait while we load your content...
Please wait while we load your content...