Design of proton-conducting Sn0.95Al0.05P2O7 with a mesoporous structure†
Abstract
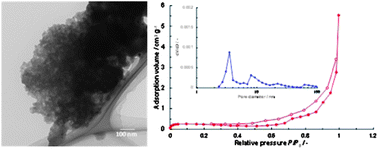
Mesopores in inorganic materials are attractive and important spaces for the development of proton conductors, because capillary condensation of water molecules occurs at relatively low relative humidity (RH), allowing for fast transport of protons without excessive humidification. Most such mesoporous inorganic compounds are constructed of silica and titania. In this study, we adopt SnP2O7 and Sn0.95Al0.05P2O7 as mesoporous frameworks. Since these pyrophosphates in themselves are proton-conducting materials, there may be synergy between the framework and mesopore spaces. Mesoporous SnP2O7 or Sn0.95Al0.05P2O7 was synthesized through a reaction between mesoporous SnO2 or Sn0.95Al0.05O1.975 and phosphoric acid, respectively. At a reaction temperature of 250 °C, only the crystalline phase attributable to SnP2O7 was detected by X-ray diffraction. The obtained compounds showed hysteresis loops in the nitrogen gas adsorption/desorption isotherms, the analyses of which indicated mean pore diameters of ca. 7 nm for SnP2O7 and ca. 4 nm for Sn0.95Al0.05P2O7. Partially destroyed hexagonal mesopores were observed in transmission electron spectroscopy observations of these compounds. The proton conductivity of the mesoporous SnP2O7 was more than one order of magnitude higher than that of the nonporous SnP2O7 in the temperature range of 50–200 °C. Furthermore, the dependence of the proton conductivity on the RH was much larger for the mesoporous SnP2O7 than for the nonporous SnP2O7. Temperature-programmed desorption analysis of water vapor revealed that water molecules filling the mesopores through capillary condensation are responsible for the high proton conductivity of the mesoporous SnP2O7 at low RH. More importantly, this capillary condensation effect was enhanced by doping SnP2O7 with Al, which shifted the RH where the proton conductivity sharply increased to a lower value. The resultant proton conductivity at 150 °C reached 0.07 S cm−1 at only 14% RH.

 Please wait while we load your content...
Please wait while we load your content...