Self-assembled hybrid tinphosphonate nanoparticles with bimodal porosity: an insight towards the efficient and selective catalytic process for the synthesis of bioactive 1,4-dihydropyridines under solvent-free conditions†
Abstract
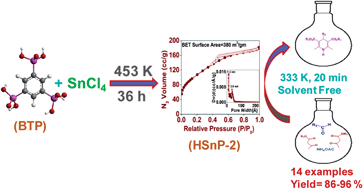
Self-assembled ordered mesoporous organic–inorganic hybrid tinphosphonate nanomaterial (HSnP-2) with crystalline framework structure has been synthesized through the reaction of benzene-1,3,5-triphosphonic acid (BTPA) and Sn(IV) chloride under hydrothermal conditions at 453 K in the absence of any structure directing agent. Powder XRD, FE-SEM, HR-TEM, N2 sorption, solid state 13C CP-MAS and 31P MAS NMR, TG-DTA analysis and FT-IR spectroscopic techniques are employed to characterize the material. The hexagonal crystal phase of the material is established through REFLEX and CELSIZ unit cell refinement programs. The calculated unit cell parameters of HSnP-2 are a = b = 17.515 Å and c = 10.254 Å. The material is composed of hexagonally ordered tiny nanocrystals of dimensions of ca. 4.0 nm and exhibits high BET surface area (380 m2 g−1), good thermal stability along with uniform supermicropores (ca. 1.3 nm) and an ordered assembly of mesopores. HSnP-2 shows outstanding catalytic activity and high recycling efficiency for the green and efficient one-pot three component coupling reaction for the synthesis of bioactive 1,4-dihydropyridines with excellent yields under solvent-free conditions. The novelty of this eco-friendly catalytic system is further manifested from the mild reaction temperature (333 K) and short reaction time (20 min), together with exclusive selectivity for the desired 1,4-dihydropyridines over 2-arylpyridines or 1,2-dihydopyridines.

 Please wait while we load your content...
Please wait while we load your content...