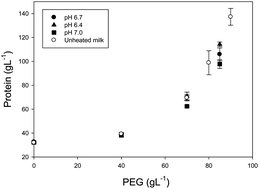
The colloidal properties of casein micelles in heated milk as a function of volume fraction are largely unknown. Heat-treatment of milk causes denaturation and unfolding of whey proteins, which then react with caseins to form a complex mixture of soluble whey protein aggregates and whey protein coated casein micelles. The distribution of the complexes between the soluble and the micellar phase depends on the pH of milk before heating. This work focused on the contribution of the heat induced complexes to the colloidal properties of concentrated casein micelles. Concentration was performed using osmotic stressing. Although there were differences in the apparent diameter after heating milk at pH 6.4, 6.8 or 7.0, redilution of the concentrated milk showed no irreversible aggregation of the casein micelles with concentration. Above 70 g L−1 protein, there was a decrease in the mean square displacement slope measured by light scattering, suggesting that above this concentration, the casein micelles were no longer free diffusing. The viscosity of the concentrated milk could be predicted using a rheological model for hard spheres assuming constant voluminosity.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...