Oxidation of ferrocenemethanol grafted to a hydrogel network through cysteine for triggering volume phase transition†
Abstract
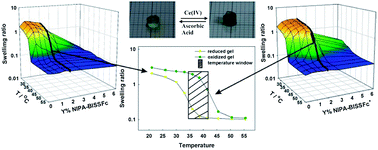
New gels based on N-isopropylacrylamide and cysteine were prepared by free radical polymerization in aqueous solutions. To attach the amino acid to the polymer chain, its α-amino group was modified with acryloyl chloride and N,N′-bisacryloylcystine was obtained. Next, the disulfide bridges were reduced to thiol groups and the attachment of ferrocenemethanol to the chains was performed. The influence of the amount of electroactive groups and their degree of oxidation on the temperature of volume phase transition was investigated. The gel formed from the polymerization solution containing 2% N,N′-bisacryloylcystine could undergo the volume phase transition triggered by changing the ferrocene state of oxidation. The temperature window of the gel sensitivity to the oxidation state of its ferrocene component was 35–40 °C. During the transition the volume of the gel could be changed even by more than an order of magnitude. Thin layers of the electroactive and thermoresponsive gel could be anchored on the surface of a glassy carbon electrode. This was achieved by electroformation of radicals at the electrode surface. The influence of the volume phase transition triggered by a temperature change on redox processes of ferrocene moieties bound to the network was examined voltammetrically.

 Please wait while we load your content...
Please wait while we load your content...