A mononuclear copper(ii) complex immobilized in mesoporous silica: an efficient heterogeneous catalyst for the aerobic oxidation of benzylic alcohols†
Abstract
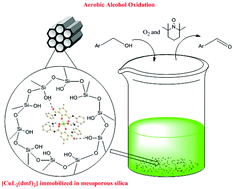
The diamide ligand 2,6-bis[(N-phenyl)amido]-4-methylphenol (HL) has been used to synthesize a mononuclear copper(II) complex, [CuL2(DMF)2] (1). The X-ray crystal structure of this complex reveals that the copper(II) center is coordinated to the two phenoxides and two amide carbonyl oxygens of the HL ligand and additionally to the amide carbonyl oxygens of two dimethylformamide molecules. This complex was also characterized using UV-Vis and EPR spectroscopy, along with thermal and electrochemical analysis. Complex 1 was then immobilized in a 2-D mesoporous silica matrix. This immobilized complex, 1a, has been characterized using UV-Vis and FT-IR spectroscopy, as well as small angle X-ray diffraction, N2 sorption and TEM. The immobilized complex was found to heterogeneously catalyze the aerobic oxidation of benzylic alcohols to the corresponding aldehydes in the presence of TEMPO, with high turnover numbers and good recyclability for up to four cycles.

 Please wait while we load your content...
Please wait while we load your content...