Photocatalytic reductive dechlorination of chlorobenzene in alkali-free aqueous alcoholic suspensions of palladium-loaded titanium(iv) oxide particles in the absence or presence of oxygen
Abstract
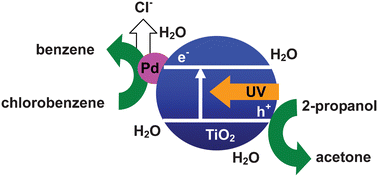
Photocatalytic reductive dechlorination of chlorobenzene in aqueous 2-propanol suspensions of palladium-loaded titanium(IV) oxide (Pd–TiO2) particles was examined in a wide range of aerated and deaerated solutions with different water and NaOH contents. In water-free 2-propanol, the presence of dissolved NaOH and removal of oxygen (O2) were critical for the dechlorination of chlorobenzene at a sufficient reaction rate and a redox balance (stoichiometry with the oxidation of 2-propanol) close to unity. In 50 vol% water–2-propanol solvent, the yield of benzene under aerated conditions was the same as that under deaerated conditions and was very close to the yield of acetone under aerated conditions, indicating that photogenerated electrons were selectively used for the reduction of chlorobenzene in the water–2-propanol solvent even in the presence of O2. The low solubility of O2 in the water–2-propanol solvents was attributed to the high selectivity of photocatalytic dechlorination. In 50 vol% water–2-propanol solvent, NaOH was unnecessary when the reaction was performed under deaerated conditions, suggesting that water played a role in removing chloride ions from the reaction site (surface of TiO2) and in suppressing the consumption of chloride ions by another side-reaction even in the absence of NaOH. Under ideal conditions, i.e., reaction in the absence of NaOH under aerated conditions, benzene was obtained in a 71% yield and a high material balance of benzene and chlorobenzene was recovered.

 Please wait while we load your content...
Please wait while we load your content...