l-Proline catalyzed highly efficient synthesis of Z-5-alkylidene cyclic sulfamidate imines: an easy access to 5-alkyl-substituted cyclic sulfamidate imines†
Abstract
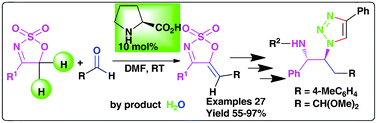
A very simple, mild and robust method for the synthesis of exclusively Z-trisubstituted 5-alkyliden cyclic sulfamidate imines via a condensation reaction of 4-aryl-5H-1,2,3-oxathiazole-2,2-dioxides with aldehydes using L-proline as an organocatalyst at room temperature is reported for the first time. In addition, the synthesis of 5-alkyl substituted cyclic sulfamidate imines, trans-β-amino-α-azido/triazole and 2-hydroxy-4-triazole-5-phenylpyrrolidine derivatives have been developed through this methodology.

 Please wait while we load your content...
Please wait while we load your content...