The effect of Si/Al ratios on the catalytic activity of CuY zeolites for DMC synthesis by oxidative carbonylation of methanol: a theoretical study
Abstract
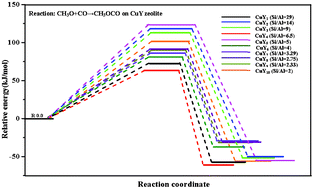
Cu-exchanged Y zeolites with different Si/Al ratios have been investigated using a density functional theory method in order to determine the effect of Si/Al ratios on the catalytic activity for producing DMC by the oxidative carbonylation of methanol. The stable structures and catalytic activity of the CuY zeolites with different Si/Al ratios are identified. In addition, CO adsorption and the effect of CH3O on CO adsorption, including the changes in the adsorption energy and stretching vibrational frequency for the adsorbed CO, are obtained. Our results indicate that the CuY zeolite with a Si/Al ratio = 6.5 has the highest catalytic activity for producing DMC by oxidative carbonylation of methanol among all the Si/Al ratios considered in this work, which is supported by the experimental facts. Finally, our results suggest that theoretical calculations can be used as a useful tool and provide good theoretical guidance for the experimental design of CuY zeolites.

 Please wait while we load your content...
Please wait while we load your content...