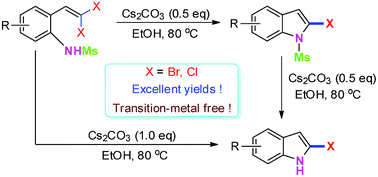
The facile synthesis of 2-bromoindoles via Cs2CO3-promoted intramolecular cyclization of 2-(gem-dibromovinyl)anilines under transition-metal-free conditions†
Abstract
2-Bromoindoles were readily prepared through a facile Cs2CO3-promoted intramolecular

 Please wait while we load your content...
Please wait while we load your content...