Near-infrared emission of novel bent-core V-shaped conjugated polymers based on the B,O-chelated azadipyrromethene structure†
Abstract
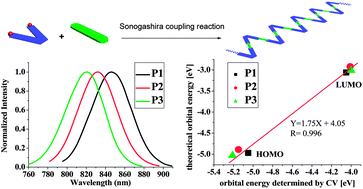
Three novel polymers with a bent-core V-shaped chain backbone, P1, P2, and P3, were synthesized by Pd-catalyzed Sonogashira coupling reaction of dibromo substituted B,O-chelated azadipyrromethene (M-1) with 2,5-diethynyl-3,4-dimethylthiophene (M-2), 3,6-diethynyl-9-octadecyl-9H-carbazole (M-3), and 1,4-diethynyl-2,5-bis(octyloxy)benzene (M-4), respectively. On varying the electron donating strength of co-monomer partners, the resulting conjugated polymers exhibited narrow near-infrared (NIR) emission over 820 nm with tunable band gaps in the range of 0.99–1.21 eV. Interestingly, there is a linearly proportional relationship between the experimentally determined energy levels (cyclic voltammetry (CV)) and the theoretically calculated ones for the HOMOs and LUMOs of these polymers. This provides us with a valuable method to predictably tune frontier molecular orbital energy levels and guide the synthesis of NIR emissive materials.

 Please wait while we load your content...
Please wait while we load your content...