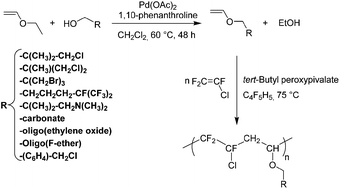
An air-stable palladium catalyst formed in situ from commercially available components efficiently catalyzed the transetherification between ethyl vinyl ether and various alcohols to lead to the corresponding original vinyl ethers in 40–84% yield in a single step. First, a series of vinyl ether monomers that bear various functional groups, such as halogenomethyl groups, amino groups, carbonate side-groups, oligo(ethylene oxide), and oligo(fluoroether) chains, were prepared and characterized by 1H and 13C NMR spectroscopy. Then, radical copolymerization of such novel vinyl ethers with chlorotrifluoroethylene (CTFE) led to alternating poly(CTFE-alt-VE) copolymers that contained various functional dangling groups. A series of copolymers with different amounts of functional groups were obtained in fair to good yield (32–81%). Their alternating structure was confirmed by elemental analysis as well as by 1H, 19F, and 13C NMR spectroscopy. Their thermogravimetric analyses under air showed decomposition temperatures at 10% weight loss (Td,10%) ranging from 214 to 324 °C. The oligo(fluoroether) chain-based copolymers appeared more thermostable than those based on other various functional groups.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...