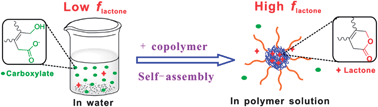
10-Hydrocamptothecin (HCPT) exists, like most of the antitumor drugs in the camptothecin (CPT) family, in the form of either lactone or carboxylate, but only the former with a closed E-ring has antitumor efficacy and the lactone fraction at physiological pH is quite low. Herein, we examined the effect of addition of an amphiphilic copolymer Pluronic P123 on HCPT in water. P123 is composed of major poly(ethylene glycol)–poly(propylene glycol)–poly(ethylene glycol) and minor diblock copolymers. The lactone fraction of HCPT in the P123 aqueous solution was increased with the copolymer concentration, and the transition P123 concentration was higher than the critical micelle concentration by about two orders of magnitude and lower than critical gelation concentration by 1.5 orders of magnitude. We anticipated that the key event was the co-assembly between drug molecules and “mature micelles” instead of either physical gelation or merely the nascent micelle formation. Such anticipation was confirmed by the fluorescence anisotropy of HCPT at various P123 concentrations. We also explicitly indicated that the enhancement of the lactone fraction was not due to a kinetic delay of the ring-opening of drugs within micelles, but a thermodynamic shift of the equilibrium between the active and inactive forms driven by drug–material interactions. Further, the equilibrium constant (Kmicelle) of HCPT within P123 micelles was determined based on analysis of partition between micelles and their medium of both the lactone and carboxylate forms, by which we afforded the so-far only equilibrium constant of the CPT-family drugs within suspended nanoparticles.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...