Photodegradation of methyl orange and photoinactivation of bacteria by visible light activation of persulphate using a tris(2,2′-bipyridyl)ruthenium(ii) complex†
Abstract
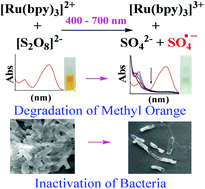
Persulphate is an emerging oxidant in the field of advanced oxidation processes for the degradation of environmentally persistent organic compounds. The present study shows that visible light activation of persulphate (2 mM) using tris(2,2′-bipyridyl)ruthenium(II) (complex 1) (1 μM) caused rapid degradation (98%) of model azo dye methyl orange (MO) (12 mg L−1) with significant mineralization (76%), and also complete inactivation of both Gram negative and positive bacteria (∼107 CFU mL−1). BacLight LIVE/DEAD assay, scanning electron microscopy and genomic DNA analysis revealed cell membrane damage and loss of chromosomal DNA, indicating oxidative stress caused to E. coli during photoinactivation. The effect of concentration of complex 1 : persulphate ratio and presence of inorganic ions (0.1 M), such as sodium hydrogen phosphate, sodium sulphate, and sodium hydrogen carbonate, on the photodegradation of MO and photoinactivation of E. coli were studied. In addition, the effect of the presence of the organic contaminant resorcinol on the photoinactivation of E. coli was also studied. Significant degradation of MO and complete inactivation of bacteria were observed in simulated ground water. The present study is the first to reveal that activation of persulphate using a visible light absorbing metal complex in aqueous media has the ability to cause degradation of organic contaminants as well as complete inactivation of bacteria.

 Please wait while we load your content...
Please wait while we load your content...