Practical oxazole synthesis mediated by iodine from α-bromoketones and benzylamine derivatives†
Abstract
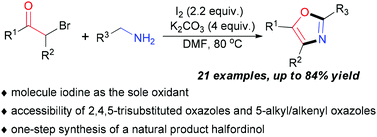
The reagent system of I2/K2CO3 could efficiently promote the oxazole synthesis from α-bromoketones and benzylamine derivatives in DMF. This method was not only suitable for 2,5-diaryl oxazole synthesis but also for 2,4,5-trisubstituted oxazole and 5-alkyl/alkenyl oxazole synthesis. Furthermore, this method was successfully applied to a one-step synthesis of a natural product halfordinol in 62% yield.

 Please wait while we load your content...
Please wait while we load your content...