Au/3DOM Co3O4: highly active nanocatalysts for the oxidation of carbon monoxide and toluene†
Abstract
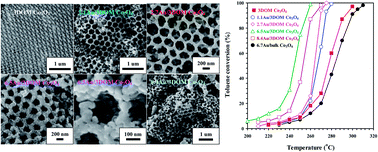
Three-dimensionally ordered macroporous Co3O4 (3DOM Co3O4) and its supported gold (xAu/3DOM Co3O4, x = 1.1–8.4 wt%) nanocatalysts were prepared using the polymethyl methacrylate-templating and bubble-assisted polyvinyl alcohol-protected reduction methods, respectively. The 3DOM Co3O4 and xAu/3DOM Co3O4 samples exhibited a surface area of 22–27 m2 g−1. The Au nanoparticles with a size of 2.4–3.7 nm were uniformly deposited on the macropore walls of 3DOM Co3O4. There were good correlations of oxygen adspecies concentration and low-temperature reducibility with catalytic activity of the sample for CO and toluene oxidation. Among 3DOM Co3O4 and xAu/3DOM Co3O4, the 6.5Au/3DOM Co3O4 sample performed the best, giving a T90% (the temperature required for achieving a conversion of 90%) of −35 °C at a space velocity of 20 000 mL g−1 h−1 for CO oxidation and 256 °C at a space velocity of 40 000 mL g−1 h−1 for toluene oxidation. The effect of water vapor was more significant in toluene oxidation than in CO oxidation. The apparent activation energies (26 and 74 kJ mol−1) over 6.5Au/3DOM Co3O4 were lower than those (34 and 113 kJ mol−1) over 3DOM Co3O4 for CO and toluene oxidation, respectively. It is concluded that the higher oxygen adspecies concentration, better low-temperature reducibility, and strong interaction between Au and 3DOM Co3O4 were responsible for the excellent catalytic performance of 6.5Au/3DOM Co3O4.

 Please wait while we load your content...
Please wait while we load your content...