Nanoscale effects of silica particle supports on the formation and properties of TiO2 nanocatalysts
Abstract
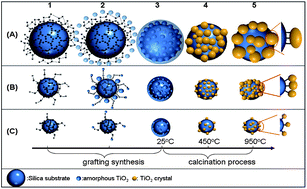
Small TiO2 crystals in the anatase phase are in high demand as photocatalysts. Stable TiO2 crystals in the anatase phase were obtained using a silica nanoparticle as a support. The focus of this study was to investigate the nanoscale effect of the silica support on the formation and properties of small anatase crystals. The experiments were carried out using powder X-ray diffraction, differential thermal analysis, transmission electron microscopy, and energy dispersion spectroscopy. The results showed that the size of the silica support played a crucial role in crystallization of TiO2 and regulation of TiO2 properties, including phase transition, crystal size, thermodynamic property and catalytic activity. A nanoscale curvature model of the spherical silica support was proposed to explain these size effects. Finally, the developed TiO2 catalysts were applied to the oxidation of methanol using a high-throughput photochemical reactor. The size effect of the silica supports on the TiO2 catalytic efficiency was demonstrated using this system.

 Please wait while we load your content...
Please wait while we load your content...