Adamantane substituted aminocyclitols as pharmacological chaperones for Gaucher disease†
Abstract
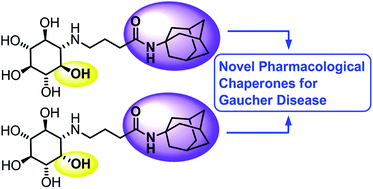
Gaucher disease (GD), resulting from deficient lysosomal enzyme β-glucosidase (GCase) activity, is the most common lysosomal storage disorder. We have previously shown that aminocyclitol derivatives displayed selective inhibition of GCase and enhanced GCase activity in N370S and L444P at very low concentrations. In the present study, we combined amino-myo-inositol and amino-scyllo-inositol cores with a hydrophobic alkyl adamantyl amide to afford novel small molecules with enhanced ability to increase GCase activity in GD lymphoblasts. The most potent inhibitor, amino-myo-inositol 2, displayed a Ki value of 250 nM in isolated enzyme. This compound produced a maximum increase of GCase activity of 64% in N370S lymphoblasts at 1 μM and 150% in L444P at 100 μM following a 3 day incubation.
 Please wait while we load your content...
Please wait while we load your content...