UV-induced atomization of gaseous mercury hydrides for atomic fluorescence spectrometric detection of inorganic and organic mercury after high performance liquid chromatographic separation
Abstract
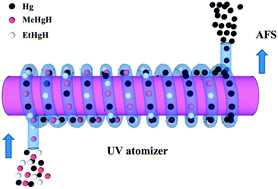
A novel, simple, low power, low temperature (<45 °C) and high efficiency atomization technique using ultraviolet (UV) radiation was proposed for the atomization of gaseous mercury hydrides for their determination by atomic fluorescence spectrometry (AFS). This technique was used for the detection of inorganic mercury (Hg2+) and organic mercury (MeHg+ and EtHg+) after high performance liquid chromatographic separation. In the proposed method, with 0.5% (m/v) KBH4 used as a reductant, inorganic mercury was reduced to elemental mercury, whereas the organic mercury species formed their respective volatile organic mercury hydrides (MeHgH and EtHgH) which were flushed to the UV atomizer for atomization and AFS detection. Under the optimized experimental conditions, the limits of detection were found to be 0.38, 0.41 and 0.56 μg L−1, and the relative standard deviations (n = 3) were 1.1%, 2.6% and 1.4% for Hg2+, MeHg+ and EtHg+, respectively. The accuracy of the proposed method was validated by analyzing a certified reference sample (fish muscle tissue) with a satisfactory analytical result, and two water samples with recoveries in the range of 93.8–97.4%.

 Please wait while we load your content...
Please wait while we load your content...