1-O-Alkyl (di)glycerol ethers synthesis from methyl esters and triglycerides by two pathways: catalytic reductive alkylation and transesterification/reduction†
Abstract
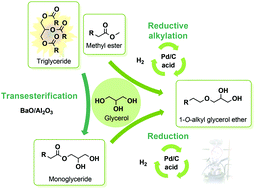
From available and bio-sourced methyl esters,
* Corresponding authors
a
Laboratoire de Catalyse, Synthèse et Environnement, Institut de Chimie et Biochimie Moléculaires et Supramoléculaires (ICBMS), CNRS. UMR5246, Université Lyon 1, Bâtiment Curien-CPE, 3° étage, 43 Bd du 11 Novembre 1918, Villeurbanne Cedex, France
E-mail:
marc.lemaire.chimie@univ-lyon1.fr
Fax: +33 472 431 408
Tel: +33 472 431 409
b
Sofiprotéol, 11, rue de Monceau, CS 60003, 75378 Paris Cedex 08, France
E-mail:
y.raoul@prolea.com
Fax: +33 147 230 288
Tel: +33 140 694 800
From available and bio-sourced methyl esters,

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
M. Sutter, W. Dayoub, E. Métay, Y. Raoul and M. Lemaire, Green Chem., 2013, 15, 786 DOI: 10.1039/C3GC36907B
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
