Abstract
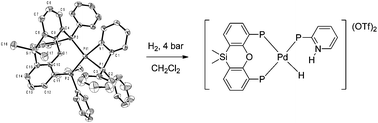
Wide bite angle diphosphine ligands were used to prepare [(diphosphine)M(2-(diphenylphosphino)pyridine)]2+ complexes (M = Pd, Pt). Except for the ligand with the largest bite angle, 2-(diphenylphosphino)pyridine coordinates in a bidentate mode leading to bis-chelate complexes. In the case of Xantphos (9,9-dimethyl-4,5-bis(diphenylphosphino)-xanthene, βn = 111°) two types of complexes are formed, in which 2-(diphenylphosphino)pyridine coordinates in a mono- or bidentate fashion, respectively. The crystal structures of three of the Pt complexes were determined. The X-ray crystal structure of [(Xantphos)-Pt(2-(diphenylphosphino)pyridine)]2+ shows that Xantphos coordinates in a tridentate P,O,P fashion. Under dihydrogen pressure, the pyridyl moiety in the platinum complexes can de-coordinate to provide a vacant coordination site at the metal center. Furthermore it can act as an internal base to assist the heterolytic cleavage of dihydrogen. The reaction yields a platinum hydride with a protonated pyridine moiety in close proximity to one another. The structure as well as the reactivity of the complexes towards dihydrogen is governed by the steric requirements of the diphosphines. The crystal structure of [(dppf)PtH(2-(diphenylphosphino)pyridinium)](OTf)2 has been determined. Palladium complexes containing DPEphos or Xantphos decompose under dihydrogen pressure. In the case of dppf slow heterolytic splitting of dihydrogen occurs to form the hydride complex [(dppf)PdH(2-(diphenylphosphino)pyridinium)](OTf)2 which contains a protonated 2-(diphenylphosphino)pyridine ligand. In solution, this compound slowly undergoes P–C bond cleavage of the 2-(diphenylphosphino)pyridine ligand to form [(dppf)Pd(PHPh2)(η1-C5H4NH)](OTf)2. When the 6-methyl-2-pyridyldiphenylphosphine ligand is used, the reaction of the palladium complex with dihydrogen is very fast and the hydride complex immediately rearranges to the diphenylphosphino compound resulting from P–C bond cleavage.
- This article is part of the themed collection: In Celebration of David Cole-Hamilton's Career in Chemistry

 Please wait while we load your content...
Please wait while we load your content...