Discrimination of cis–trans isomers by dinuclear metal cryptates at physiological pH: selectivity for fumarate vs. maleate†
Abstract
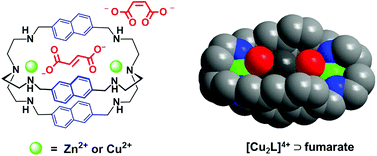
Cryptand L (L = N[(CH2)2NHCH2(2,6-C10H6)CH2NH(CH2)2]3N) and its dinuclear metal cryptates [Zn2L](NO3)4 (1) and [Cu2L](ClO4)4 (2) have been prepared, and the binding properties of the cryptates with fumarate and its cis isomer maleate were investigated using fluorescent

 Please wait while we load your content...
Please wait while we load your content...