Thermodynamics of complexation reactions of borate and phenylboronate with diol, triol and tetritol†
Abstract
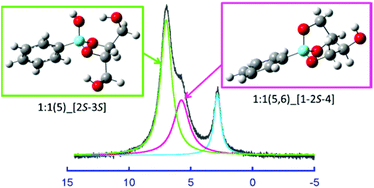
In order to fully elucidate the structures and stabilities of borate and phenylboronate complexes with polyols, the complexations with 1,2-ethanediol, 1,3-propanediol, 1,2,3-propanetriol, erythritol and threitol were investigated by NMR spectroscopy and DFT calculations. For the borate, 1 : 1 complexes binding with a ligand in a monodentate manner and 1 : 2 complexes binding with one ligand in an α,β- or α,γ-bidentate manner and the other in a monodentate manner were identified for the first time besides the well-known 1 : 1 and 1 : 2 complexes with five- and/or six-membered chelate rings. In addition, the formations of 1 : 1 α,β,δ-tridentate complexes with tetritol were confirmed. For the phenylboronate, the bidentate and tridentate complexes with 1 : 1 stoichiometries were identified. These complexations were enthalpy-driven processes accompanied by a negative entropic contribution. Linear enthalpy–entropy compensation relationships were observed for the stepwise complexations of borate and phenylboronate to form a five- or six-membered chelate ring. The TΔS° vs. ΔH° plots gave three different straight lines in parallel with one another depending on the number of OH groups outside the chelate ring. For the second intramolecular chelation to form the α,β,δ-tridentate complex from the α,β- or α,γ-bidentate complex with tetritol, the TΔS° vs. ΔH° plots gave different straight lines for the borate and phenylboronate, being more favorable in enthalpy for the phenylboronate.

 Please wait while we load your content...
Please wait while we load your content...