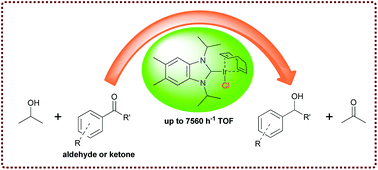
Two new [Ir(NHC)(COD)Cl] (NHC = N-heterocyclic carbene; COD = 1,5-cyclooctadiene) iridium complexes (2a,b) have been prepared by the reaction of [Ir(COD)Cl]2 with in situ prepared NHC–Ag carbene transfer agents in dichloromethane at ambient temperature. They have been fully characterized by 1H, 13C NMR, and elemental analysis. X-ray diffraction studies on single crystals of 2a and 2b confirm the square planar geometry. Complexes of type [Ir(NHC)(CO)2Cl] [NHC = 1,3-diisopropyl(5,6-dimethyl)benzimidazol-2-ylidene] 3 were also synthesized to compare σ-donor/π-acceptor strength of NHC ligands. Transfer hydrogenation (TH) reactions of various aldehydes and ketones have been studied using complexes 2a and 2b as catalysts. The 5,6-dimethyl substituted iridium complex (2b) showed the highest catalytic activity for the TH reaction.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...