Be squared: expanding the horizon of squaric acid-mediated conjugations
Abstract
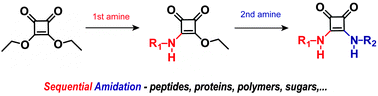
Squaric acid diesters can be applied as reagents to couple two amino-functional compounds. Consecutive coupling of two amines allows the synthesis of asymmetric squaric acid bisamides with either low molecular weight compounds but also biomolecules or polymers. The key feature of the squaric acid diester mediated coupling is the reduced reactivity of the resulting ester-amide after the first amidation step of the diester. This allows the sequential amidation and generation of asymmetric squaramides with high selectivity and in high yields. This article gives an overview of the well-established squaric acid diester mediated coupling reactions for glycoconjugates and presents recent advances that aim to expand this very versatile reaction protocol to the modification of peptides and proteins.

 Please wait while we load your content...
Please wait while we load your content...