LixFeF6 (x = 2, 3, 4) battery materials: structural, electronic and lithium diffusion properties†
Abstract
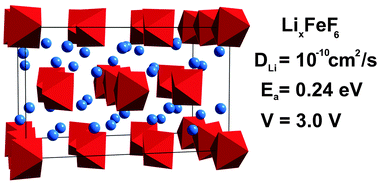
Lithium iron fluoride materials have attracted recent interest as cathode materials for lithium ion batteries. The electrochemical properties of the high energy density LixFeF6 (x = 2, 3, 4) materials have been evaluated using a combination of potential-based and DFT computational methods. Voltages of 6.1 V and 3.0 V are found for lithium intercalation from Li2FeF6 to α-Li3FeF6 and α-Li3FeF6 to Li4FeF6 respectively. The calculated density of states indicate that Li2FeF6 possesses metallic states that become strongly insulating after lithium intercalation to form α-Li3FeF6. The large energy gain associated with this metal–insulator transition is likely to contribute to the associated large voltage of 6.1 V. Molecular dynamics simulations of lithium diffusion in α-Li3FeF6 at typical battery operating temperatures indicate high lithium-ion mobility with low activation barriers. These results suggest the potential for good rate performance of lithium iron fluoride cathode materials.

 Please wait while we load your content...
Please wait while we load your content...