Computational study of peptide bond formation in the gas phase through ion–molecule reactions†
Abstract
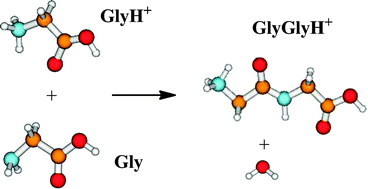
A computational study of
* Corresponding authors
a
Departamento de Química Física y Química Inorgánica, Facultad de Ciencias, Universidad de Valladolid, 47011 Valladolid, Spain
E-mail:
alargo@qf.uva.es
b Departamento de Química Orgánica, Escuela de Ingenierías Industriales, Universidad de Valladolid, 47011 Valladolid, Spain
c LAMBE - Laboratoire Analyse et Modelisation pour la Biologie et l'Environnement, Universite d'Evry val d'Essonne, UMR 8587, Blvd F. Mitterrand, Bat Maupertuis, 91025 Evry, France
A computational study of

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
P. Redondo, H. Martínez, Á. Cimas, C. Barrientos and A. Largo, Phys. Chem. Chem. Phys., 2013, 15, 13005 DOI: 10.1039/C3CP51535D
To request permission to reproduce material from this article, please go to the Copyright Clearance Center request page.
If you are an author contributing to an RSC publication, you do not need to request permission provided correct acknowledgement is given.
If you are the author of this article, you do not need to request permission to reproduce figures and diagrams provided correct acknowledgement is given. If you want to reproduce the whole article in a third-party publication (excluding your thesis/dissertation for which permission is not required) please go to the Copyright Clearance Center request page.
Read more about how to correctly acknowledge RSC content.
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
