Encapsulation of LiNi0.5Co0.2Mn0.3O2 with a thin inorganic electrolyte film to reduce gas evolution in the application of lithium ion batteries†
Abstract
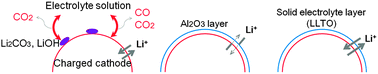
Emission of gas at charged states of lithium ion batteries (LIBs) is a significant problem because it causes swelling and deformation of LIBs. In this study, the gas generation mechanism is investigated and preventative measures are developed. Decomposition of the electrolyte solution related to residual lithium compounds on the surface of LiNi0.5Co0.2Mn0.3O2 (NCM523) and to the structural change in the cathode active material is investigated as two main mechanisms of gas generation in LIB cells. NCM523 particles are encapsulated in a continuous lithium lanthanum titanium oxide (LLTO) thin layer to inhibit gas-generating reactions. The LLTO layer fixes free lithium of the cathode surface and effectively suppresses side reactions between the charged cathode active material and the electrolyte solution, resulting in substantial reduction of the gas generation. In addition, LLTO-coated NCM523 shows improved capacity retention without any loss of capacity or rate performance because LLTO is a good conductor of Li ions.

 Please wait while we load your content...
Please wait while we load your content...