Electrochemical dealloying of Al2(Au,X) (X = Pt, Pd, PtPd, Ni, Co and NiCo) alloys in NaCl aqueous solution†
Abstract
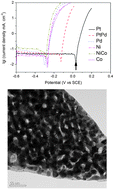
The electrochemical dealloying of rapidly solidified Al2(Au,X) (X = Pt, Pd, PtPd, Ni, Co and NiCo) precursors in a 1.0 M NaCl aqueous solution has been systematically investigated using electrochemical measurements including open-circuit measurement, potentiodynamic polarization and potentiostatic polarization, and microstructural analysis. The results show that the kind of alloying element(s) has a significant influence on the open-circuit and corrosion potentials of the rapidly solidified Al2(Au,X) precursors. The bulk dealloying of the Al2(Au,X) precursors is affected by the kind of alloying element(s) and also sensitive to the applied potential (or overpotential). The addition of Ni or/and Co easily leads to

 Please wait while we load your content...
Please wait while we load your content...