Effects of Au fraction on the morphology and stability of Au–Ag–Cu trimetallic particles prepared using a polyol method†
Abstract
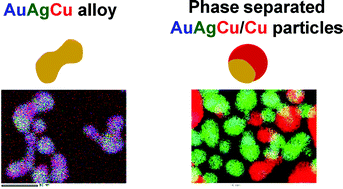
AuAgCu alloy and phase-separated AuAgCu/Cu nanoparticles were prepared by reducing mixtures of Au(OAc)3, AgNO3, and Cu(OAc)2·H2O in ethylene glycol (EG) in the presence of poly(vinylpyrrolidone) (PVP) at 175 °C. At Au(OAc)3 : AgNO3 : Cu(OAc)2·H2O molar ratios of 1 : 1 : 1, short string and peanut types of AuAgCu alloy nanoparticles were formed after 2.5–40 min heating. For Au(OAc)3 : AgNO3 : Cu(OAc)2·H2O molar ratios of 0.5 : 1 : 1 and 0.1 : 1 : 1, peanut-type or spherical AuAgCu alloy core Cu shell nanoparticles denoted as AuAgCu@Cu were produced after 5 min heating. They changed to a mixture of AuAgCu alloy and phase-separated AuAgCu/Cu nanoparticles after 17.5–37 min heating. The growth mechanisms of AuAgCu, AuAgCu@Cu, and AuAgCu/Cu particles were examined using TEM energy dispersed X-ray spectroscopic (EDS), XRD, and ultraviolet (UV)-visible (Vis)-near infrared (NIR) extinction spectral data. The time dependence of UV-Vis-NIR spectral data indicated that AuAgCu and AuAgCu/Cu nanoparticles retain high stability including anti-oxidation properties for a long time after preparation.

 Please wait while we load your content...
Please wait while we load your content...