Water-mediated supramolecular architecture of Co(iii)–phenanthroline complexes: organizational control to 2D-layers and 3D-square cavities through substituted aryl carboxylate anions†
Abstract
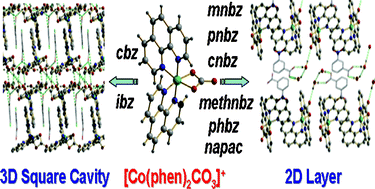
To explore the variation in the supramolecular architecture of cobalt(III)–1,10-phenanthroline–aryl carboxylate complexes in tune with ![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , Z = 2, n = 4; 1), 4-nitrobenzoate (P
, Z = 2, n = 4; 1), 4-nitrobenzoate (P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , Z = 2, n = 5; 2), 2-chloro-4-nitrobenzoate (P
, Z = 2, n = 5; 2), 2-chloro-4-nitrobenzoate (P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , Z = 2, n = 5; 3), 3-methyl-4-nitrobenzoate (P
, Z = 2, n = 5; 3), 3-methyl-4-nitrobenzoate (P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , Z = 2, n = 5; 4), 4-chlorobenzoate (P21/c, Z = 4, n = 7; 5), 2-iodobenzoate (P
, Z = 2, n = 5; 4), 4-chlorobenzoate (P21/c, Z = 4, n = 7; 5), 2-iodobenzoate (P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , Z = 2, n = 7; 6), 2-phenoxybenzoate (P
, Z = 2, n = 7; 6), 2-phenoxybenzoate (P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , Z = 2, n = 8; 7) and 2-napthleneacetate (P21/c, Z = 4, n = 7; 8) and were unequivocally characterized by physicochemical methods, spectroscopic techniques and
, Z = 2, n = 8; 7) and 2-napthleneacetate (P21/c, Z = 4, n = 7; 8) and were unequivocally characterized by physicochemical methods, spectroscopic techniques and

 Please wait while we load your content...
Please wait while we load your content...