Efficient asymmetric synthesis of lamivudine via enzymatic dynamic kinetic resolution†
Abstract
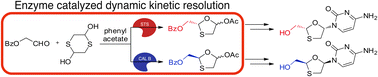
The anti-HIV nucleoside lamivudine was asymmetrically synthesized in only three steps via a novel surfactant-treated subtilisin Carlsberg-catalyzed dynamic kinetic resolution protocol. The enantiomer of lamivudine could also be accessed using the same protocol catalyzed by Candida antarctica lipase B.

 Please wait while we load your content...
Please wait while we load your content...