Diatomaceous earth supported liquid extraction and LC-MS/MS determination of elvitegravir and ritonavir in rat plasma: application to a pharmacokinetic study
Abstract
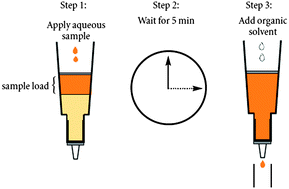
A rapid, sensitive and selective bio-analytical method was developed for determination of elvitegravir and ritonavir in rat plasma by high-performance liquid chromatography–tandem mass spectrometry. Sample preparation was based on diatomaceous earth supported liquid extraction using dichloromethane to extract elvitegravir and ritonavir from rat plasma. Chromatographic separation was performed on a Waters Symmetry C18 column (4.6 × 250 mm, 5 μm) using 20 mM ammonium formate containing 0.05% trifluoroacetic acid and acetonitrile (35 : 65 v/v) for isocratic elution and mass detection. Calibration curves were linear with regression coefficients greater than 0.997 over the concentration range of 2–5000 ng mL−1 for elvitegravir and ritonavir. Absolute mean recoveries were in the range of 87.31 to 92.18% and no significant interferences were observed at the retention times of the elvitegravir, ritonavir and internal standard. The method exhibited good intra- and inter-day performance in terms of 1.55–7.47% precision and 0–5% accuracy. The developed method has been successfully applied to the pharmacokinetic study of elvitegravir in healthy rats following a single oral administration of a 15 mg kg−1 dose boosted with 5 mg kg−1 ritonavir.

 Please wait while we load your content...
Please wait while we load your content...