Development and validation of LC–MS/MS method for the quantitation of lenalidomide in human plasma using Box–Behnken experimental design
Abstract
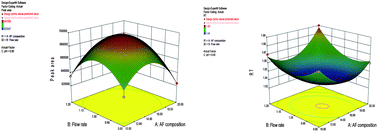
For the determination of lenalidomide using carbamazepine as an internal standard, an ultra-fast stability indicating LC–MS/MS method was developed, validated and optimized to support clinical advancement. The samples were prepared by solid-phase extraction. The calibration range was 2–1000 ng mL−1, for which a quadratic regression (1/x2) was best fitted. The method was validated and a 32 factorial was employed using Box–Behnken experimental design for the validation of robustness. These designs have three factors such as mobile phase composition (X1), flow rate (X2) and pH (X3) while peak area (Y1) and retention time (Y2) were taken as response. This showed that little changes in mobile phase and flow rate affect the response while pH has no affect. Lenalidomide and carbamazepine were stable in human plasma after five freeze thaw cycles, at room temperature for 23.7 h, bench top stability for 6.4 h. This method competes with all the regulatory requirements for selectivity, sensitivity, precision, accuracy, and stability for the determination of lenalidomide in human plasma, as well as being highly sensitive and effective for the pharmacokinetic and bioequivalence study of lenalidomide.

 Please wait while we load your content...
Please wait while we load your content...