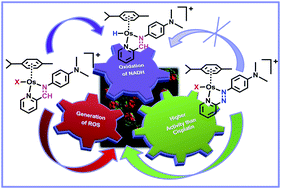
A wide variety of steric and electronic features can be incorporated into transition metal coordination complexes, offering the prospect of rationally-designed therapeutic agents with novel mechanisms of action. Here we compare the chemical reactivity and anticancer activity of organometallic OsII complexes [Os(η6-arene)(XY)Z]PF6 where arene = p-cymene or biphenyl, XY = N,N′-chelated phenyliminopyridine or phenylazopyridine derivatives, and Z = Cl or I. The X-ray crystal structure of [Os(η6-p-cym)(Impy-OH)I]PF6·0.5CH2Cl2·H2O (Impy-OH = 4-[(2-pyridinylmethylene)amino]-phenol) is reported. Like the azopyridine complexes we reported recently (Dalton Trans., 2011, 40, 10553–10562), some iminopyridine complexes are also potently active towards cancer cells (nanomolar IC50 values). However we show that, unlike the azopyridine complexes, the iminopyridine complexes can undergo aquation, bind to the nucleobase guanine, and oxidize coenzyme nicotine adenine dinucleotide (NADH). We report the first detection of an Os-hydride adduct in aqueous solution by 1H NMR (−4.2 ppm). Active iminopyridine complexes induced a dramatic increase in the levels of reactive oxygen species (ROS) in A549 lung cancer cells. The anticancer activity may therefore involve interference in the redox signalling pathways in cancer cells by a novel mechanism.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...