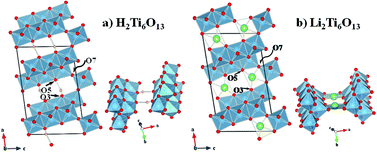
The hexatitanate H2Ti6O13 is obtained by a simple successive Na+/Li+/H+ ion exchange of Na2Ti6O13. The crystal structure of H2Ti6O13 was solved from both synchrotron and neutron powder diffraction. H2Ti6O13 crystallizes in the monoclinic space group C2/m, with a = 14.6702(3) Å; b = 3.7447(1) Å; c = 9.2594(2) Å; β = 96.941(2)°. The monoclinic symmetry of the [Ti6O13]2− framework is preserved during the exchange reaction. When compared to the positions of Na and Li in Na2Ti6O13 and Li2Ti6O13, the position of the proton is shifted towards the O3 atomic position, where it forms a covalent O–H bond. The vicinity of the proton to the O5 atom across the tunnel allows for the formation of a classical (asymmetric) hydrogen bond. H2Ti6O13 has been tested as a Li insertion material to assess its use as an electrode in lithium rechargeable batteries. It reacted irreversibly with ca. 6 Li ions per formula unit at an average voltage of 1.5 V vs. Li+/Li, with a specific discharge capacity of 315 mA h g−1. However, after first discharge, a reversible specific capacity of 170 mA h g−1 was developed. H2Ti6O13 then yielded a higher reversible specific capacity than Na2Ti6O13 and comparable to Li2Ti6O13. Besides structural details, IR spectroscopy has been used to further assess possible reaction mechanisms pointing to the transformation of H2Ti6O13 to Li2Ti6O13 when reacting with the very first two lithium ions.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...