π-Cation interactions as the origin of the weak absorption at 532 nm observed in tryptophan-containing polypeptides†
Abstract
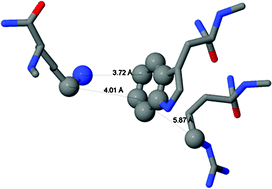
We have previously reported that bovine serum albumin (BSA) and other proteins that do not contain prosthetic groups exhibited a weak light absorption in the visible, only detectable by pulsed laser-induced optoacoustic spectroscopy (LIOAS). Human serum albumin (HSA) exhibited signals 25% higher than those observed with BSA. Signals comparable to those obtained with BSA were observed with poly(L-Trp, L-Lys), poly(L-Trp, L-Arg) or poly(L-Trp, L-Orn) at pH 7.0. No signals were obtained when tryptophan was replaced by other amino acids or when free tryptophan or the tripeptide Lys-Trp-Lys was assayed (pH 7.0). Tryptophan in HCl 5 N produced LIOAS signals similar to those produced by tryptophan-containing copolymers. Moreover, the absorption peak could be observed in a UV-VIS spectrophotometer. Therefore, the LIOAS signals obtained with BSA, HSA, and tryptophan-containing random copolymers may be attributed to a new transition of the indole moiety of their tryptophan residues when “protonated”. Tryptophan residues of proteins are known to participate in π-cation interactions, which are important in protein stability and function. As a matter of fact, HSA and BSA contain an internal tryptophan in close proximity to lysine and arginine residues and therefore suitable for π-cation interactions. The strength of this type of interaction strongly depends on distances and relative orientations of both amino acid residues. Accordingly, these interactions should be highly sensitive to conformational changes. Based on preliminary results that have shown that LIOAS signal at 532 nm depended on the aggregation state of BSA and/or on the oxidation state of its Cys-34, we postulate that the LIOAS signal observed with proteins and tryptophan-containing polypeptides are related to Trp-Lys or Trp-Arg interactions and that the intensity of the signal depends on the strength of such interactions.
- This article is part of the themed collection: In honour of Professor Kurt Schaffner

 Please wait while we load your content...
Please wait while we load your content...