A search for BACE inhibitors reveals new biosynthetically related pyrrolidones, furanones and pyrroles from a southern Australian marine sponge, Ianthella sp.†
Abstract
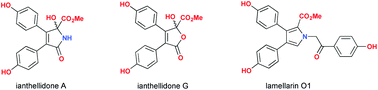
Fractionation of a southern Australian marine sponge, Ianthella sp., yielded sixteen metabolites including a new class of pyrrolidone, ianthellidones A–F (1–6), a new class of furanone, ianthellidones G–H (7–8), new and known lamellarins, lamellarins O1 (9), O2 (10), O (11) and Q (12), plus the known 4-hydroxybenzaldehyde (13), 4-hydroxybenzoic acid (14), 4-methoxybenzoic acid (15) and ethyl 4-hydroxybenzoate (16). Structures for all Ianthella metabolites were determined by detailed spectroscopic analysis, supported by a plausible biosynthetic relationship. The ianthellidones were non-cytotoxic towards two human colon cancer cell lines (SW620 and SW620 Ad300), as well as Gram +ve and Gram −ve bacteria, and a fungus. Ianthellidone F (6) and lamellarins O2 (10) and O (11) displayed modest BACE inhibitory properties (IC50 > 10 μM), while lamellarin O1 (9) was more potent (IC50 < 10 μM). Lamellarin O (11) exhibited modest cytotoxicity towards SW620 and SW620 Ad300 cell lines (IC50 > 22 μM), was an inhibitor of the multi-drug resistance efflux pump P-glycoprotein, and displayed selective growth inhibitory activity against the Gram +ve bacterium Bacillus subtilis (ATCC 6633) (IC50 2.5 μM).

 Please wait while we load your content...
Please wait while we load your content...