Developing asymmetric iron and ruthenium-based cyclone complexes; complex factors influence the asymmetric induction in the transfer hydrogenation of ketones†
Abstract
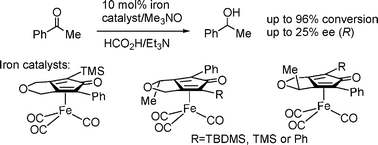
The preparation of a range of asymmetric iron and ruthenium-cyclone complexes, and their application to the asymmetric reduction of a

 Please wait while we load your content...
Please wait while we load your content...