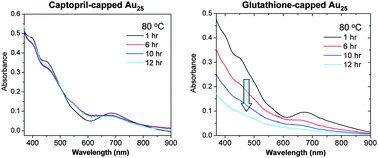
This work was motivated by the unsatisfactory stability of Au25(SG)18 in solution under thermal conditions (e.g. 70–90 °C for DNA melting). Thus, we searched for a better, water-soluble thiol ligand. Herein, we report a one-pot synthesis and investigation of the stability and optical properties of captopril (abbreviated Capt)-protected Au25(Capt)18 nanoclusters. The Au25(Capt)18 (anionic, counterion: Na+) nanoclusters were formed via size focusing under ambient conditions. Significantly, Au25(Capt)18 nanoclusters exhibit largely improved thermal stability compared to the glutathione (HSG) capped Au25(SG)18. Both Au25(Capt)18 and Au25(SG)18 nanoclusters show fluorescence centered at ∼700 nm. The chiral ligands (Capt, SG, as well as chirally modified phenylethanethiol (PET*)) give rise to distinct chiroptical features. The high thermal stability and distinct optical properties of Au25(Capt)18 nanoclusters render this material quite promising for biological applications.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?

 Please wait while we load your content...
Please wait while we load your content...